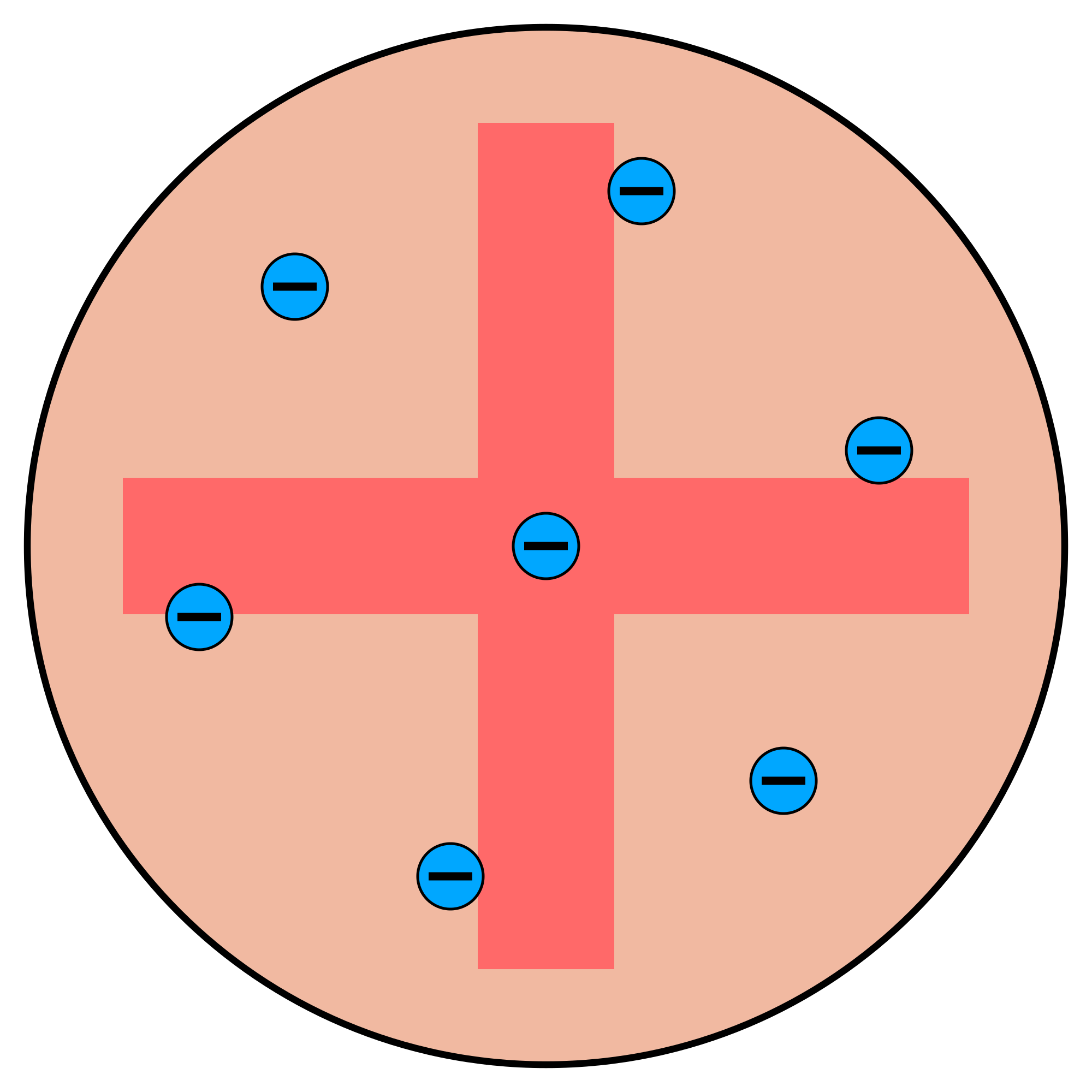
Before Geiger and Marsden’s experiment – plum pudding model of atom existed
If atoms were like that, the alpha particles which have a positive charge would pass straight through the gold foil without any deflection as the negative charge via the electrons is negligible and and there is no concentration of positive charge that would cause the positive alpha particles to repel and deflect
This apparatus was used to test the hypothesis of the plum pudding model of atom. A thin beam of alpha particle was shot through the very thing gold foil. The inside of the equipment was coated with zinc sulphide photographic film which would change color if alpha particles hit it. This allowed the scientist to see where the alpha particles went after they hit the gold foil.
A vacuum was created inside the apparatus to remove all the air so that the alpha particles are not obstructed by the air.
When the experiment was carried out, Geiger and Marsden discovered that not all alpha particles went straight through the thin gold leaf. Some of the deflected a little bit and very few came back to the source. With this information the concluded that there was a concentration of positive charge in the atoms which was causing the positively charged alpha particles to repel and delect back. The also concluded that only a small part of the atom had concentration of positive charge and the rest was empty space as the detected particles were very few and most other particles went straight through.
The deflection of alpha particles increases if we reduce the speed of the particles or increase the positive charge in the atoms, by having an element that has more protons.